How do industrial processes use equilibrium?
Industrial processes use dynamic equilibrium to get the most product from reactants. They also use Le Chatalier's Principle and other principles that use reaction kinetics to achieve the highest amount of yield by obtaining the best reaction conditions.
Lime Production from Limestone

- Lime and carbon dioxide is created from calcium carbonate
CaCO3(s) ⇌CaO(s) + CO2(g) (ΔH = +178 kJ mol-1) - The forward reaction of the equation is endothermic, so the 178kJ of heat energy will be absorbed when every mole of CaO if formed
- The formation of CaO is favored by the condition of high temperature-- reaction is not actually capable of being done until temps of at least 900ᣞC is reached, at 1000ᣞC the equilibrium of the limekiln is on the right hand side
- One mole of gas will be formed in this process, so the net of the moles increase in lime formation---by decreasing the pressure, this will allow for more gas molecules to be formed, or more CO2, which results in more lime
Synthesis of Ammonia Haber Process

Ammonia is synthesized (made) in the chemical process by reacting nitrogen gas with hydrogen gas. The three raw materials that are used include air, higher hydrocarbons such as coal-coke, or methane; and water. Nitrogen is obtained through two principle methods:
- Liquefaction-this occurs by turning air into liquid air, by cooling it then placing it under very high pressure. The liquid air is then distilled at low temps to separate oxygen (used for welding, and hospitals), nitrogen (making of ammonia), and noble gases (argon for light bulbs, and helium for balloons)
- Burning natural gas in air-separates nitrogen from water and the products from CO2 combustion
The main source of hydrogen is through gas that is created (synthesized), even though this type of gas (syngas) is made up of both hydrogen and carbon dioxide. It is made using three methods, hydrogen can also be obtained from being a by-product of oil cracking, as well as the electrolysis of brine. In the past, synthesis gas was made by using steam and white coke, where the steam would pass over the hot coke at temps reaching 800-1200 degrees Celsius. Coke being made up of mainly carbon, is made by heating coal up through high temperatures. This method is still used in many places throughout the world, especially if coal is available instead of oil, this process is also cheaper. The chemical equation is C(s) + H2O(g) CO(g) + H2(g) (ΔH = +131 kJ mol-1). By having high temperatures, this will favor the endothermic production of hydrogen which will increase the rate of the reaction by a lot. There is little reaction of water and carbon at low temperatures. The reaction will also be carried out at normal pressure, as higher pressures would favor the reverse reaction (1 gas mol ← 2 gas mol).
It will also be produced using these methods as well: - Partial oxidization of methane through the reaction of oxygen in the air:
Chemical equation: CH4(g) + 1/2O2(g) ==> CO(g) + 2H2(g) (ΔH = -36 kJ mol-1). Both low temperatures and pressure will favor this reaction. This reaction can also be done with 100% conversion at atmospheric pressure (low pressure) at 800-1000 degree Celsius by using cobalt or nickel based catalysts, and if the conditions of the reaction are controlled carefully then very little water or carbon dioxide will be formed.Though high temperatures are used (with catalysts) to break the bonds of C-H and O=O in the molecules that are the reactants and to get a good rate of the reaction, the increase of the entropy causes the moles of gas to double which then will favor the right side.This reaction will then also produce the 1:2 ratio that is needed for methanol production between CO and H2. - Steam-methane reforming:
Bosch-Haber Process for the Synthesis of Ammonia
The formation of ammonia is an exothermic reaction, as heat energy is given off to its surroundings, which actually gives off 46 kJ of heat released per mole of ammonia formed. Chemical equation: N2(g) + 3H2(g) 2NH3(g) (ΔH = -92 kJ mol-1. It takes 4 moles of the reactant gases to form 2 moles of the product gas, therefore having a decrease in the amount of gas molecules during the formation of ammonia. By applying the equilibrium rules, the formation of ammonia is favored by
* The word 'yield' basically means how much product you get compared to the maximum theoretical amount possible if the reaction becomes 100% complete. Conditions to produce the most amount of ammonia are:
- Using high pressure because you are going from 4 to 2 gas molecules (the high pressure also speeds up the reaction because it increases the concentration of the gas molecules) and allows a there to be a larger bulk rate flow of material through the reactor. However when there is high pressure the conditions of performing this reaction becomes more dangerous and costs more money as health and safety measures have to be taken
- Carrying out the reaction at a low temperature, because it is an exothermic reaction favored by low temperature, but this may produce too slow a rate of reaction
* The word 'yield' basically means how much product you get compared to the maximum theoretical amount possible if the reaction becomes 100% complete. Conditions to produce the most amount of ammonia are:
- Occurs in higher temperatures (400-500) with an iron oxide catalyst
- Catalysts do not affect the position of the equilibrium, only makes the reaction go faster
- Reaction has to occur at higher temps because nitrogen is a stable molecule, therefore not making it very reactive, and the process of using a high percent yield of ammonia with low pressures would be too slow to have any economic benefits.
For this process to be economically advantageous, high temperatures must be used which will produce less ammonia, however it does this faster than with low temperatures. From the process, the gases will come out of the iron catalyst chamber, and then cooled under high pressure, where the ammonia then liquefies.
Sulfuric Acid Production using the Contact Process

- Sulfur dioxide is mixed with air having a stoichiometric ratio of volume/mole ratio of SO2: O2 is 2:1, the mixture is used with a catalyst of vanadium oxide (V2O5) at a fairly high temp of 450 and this occurs at a pressure of 1-2atm. In the reactor the sulfur dioxide is oxidized, the chemical equation then being 2SO2(g) + O2(g) ⇋ 2SO3(g) (ΔH = -196 kJ mol-1), making it an exothermic reaction. This forms SO3 (sulfur trioxide), and the equilibrium moves to the right for these reasons:
- A higher temperature is used even though the reaction is exothermic, this is because it will favor the reverse reaction (right side to left side) by using the equilibrium energy change rule→ by increasing the temperature the equilibrium will shift to the endothermic direction
- High pressure is used, however only a small increase in pressure will be used to give high yield of sulfur trioxide, this is due to the fact the the right side is energetically quite favorable as it is exothermic and has a high Kp (equilibrium constant)
- By having the catalyst the reaction occurs quite fast, and allows the reaction to occur at not too high of temperatures
- Reactor beds (type of device that can be used to carry out many different multiphase chemical reactions) used to make sure that there is the highest percentage of conversion, and heat exchange systems are used to control the temperatures as well as the preheated gases that are coming into the reaction (reactant gases)
Methanol Production

Methanol is an alcohol which is used in fuels, making methyl esters, and creating urea-formaldehyde resin glues through the oxidation of methanol. It is produced from the synthesis of CO + H2 gas. The chemical equation is CO(g) + 2H2(g) --> CH3OH(g) (ΔH = -90 kJ mol-1). The reaction takes place at a temperature of 250 degrees using a catalyst (Cu-ZnO-Al2O3), at a pressure of about 55-100atm. In theory, a high pressure is used and a low temperature (3 gas moles→ 1 gas mole). In practice however, the reaction takes place at a high pressure and somewhat high temperature with a catalyst to get a good amount of yield.
Solvay Process of Sodium Carbonate
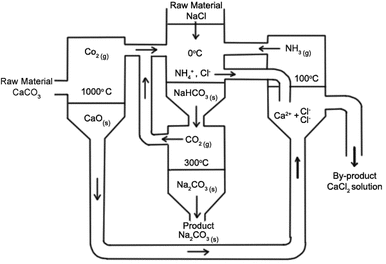
Is the method of making sodium carbonate from sodium chloride, ammonia, and calcium carbonate (limestone). This process was first used in the attempt to find a use for ammonia by Ernest Solvay, as ammonia was produced as a by-product in the coke industry. They decided to use this process and expand on it because sodium carbonate made the production of soap and glass less expensive. The overall process chemical equation is CaCO3(s) + 2NaCl (aq) --> Na2CO3(aq)+CaCl2(aq). Though ammonia is involved, it does not appear in the equation because it is recovered.
The reaction is carried out in a number of steps as calcium carbonate cannot react with sodium chloride, these steps are:
1) Brine purification-Salt water (brine) is pumped into shallow ponds, where the water is evaporated by the sun leaving salt. This is a mixture of calcium and magnesium salts as well as sodium chloride. The Ca and Mg ions must be removed. The calcium salts are made into solids by the adding sodium carbonate. Its chemical equation looks like this Ca2+(aq) + CO3(aq) + CaCO3(s). The magnesium salts are also then made into solids by the adding sodium hydroxide, which looks like this Mg2+(aq) + 2OH–(aq) -->Mg(OH)2(s). A flocculant is added, and this is what causes suspended particles to clump together and fall out of solution, the solids are then taken off the brine.
2) Production of hydrogen carbonate and sodium carbonate- Calcium carbonate is heated in a kiln to form carbon dioxide and calcium oxide (CaCO3(s)--> CO2(g) + CaO(s) ). The calcium oxide produced is removed to be used for ammonia recovery, and the coke that is present in the kiln produces more carbon dioxide when it is heated. The heat that is emitted causes the calcium carbonate to decompose. Ammonia will then be dissolved in NaCl, a purified brine, and the carbon dioxide is then dissolved in this solution. Chemical equation NaCl(aq) + NH3(g) + H2O(l) + CO2(g) --> H4Cl(aq) + NaHCO3(aq). The reaction is done at a low temperature (0 degrees) so that the sodium hydrogen carbonate precipitates out. Mixture is then filtered, and the sodium hydrogen carbonate is washed then dried and used to make sodium carbonate. After this, the ammonium chloride filtrate is sent to an ammonia recovery plant where the ammonia is recovered and reused. Sodium hydrogen is heated at about 300 degrees and then it decomposes to sodium carbonate and carbon dioxide sodium carbonate is removed then sold, the left over carbon dioxide is reused.
3) Ammonia Recovery- Calcium oxide (from the first step) is dissolved in water to form calcium hydroxide. CaO(s) + H2O(l) --> Ca(OH)2(aq). Ammonium chloride is reacted with this calcium hydroxide which forms calcium chloride and ammonia.The ammonia is reused, but calcium chloride is waste. 2NH4Cl(aq) + Ca(OH)2(s) --> 2NH3(g) + CaCl2(aq) + 2H2O(l)
The reaction is carried out in a number of steps as calcium carbonate cannot react with sodium chloride, these steps are:
1) Brine purification-Salt water (brine) is pumped into shallow ponds, where the water is evaporated by the sun leaving salt. This is a mixture of calcium and magnesium salts as well as sodium chloride. The Ca and Mg ions must be removed. The calcium salts are made into solids by the adding sodium carbonate. Its chemical equation looks like this Ca2+(aq) + CO3(aq) + CaCO3(s). The magnesium salts are also then made into solids by the adding sodium hydroxide, which looks like this Mg2+(aq) + 2OH–(aq) -->Mg(OH)2(s). A flocculant is added, and this is what causes suspended particles to clump together and fall out of solution, the solids are then taken off the brine.
2) Production of hydrogen carbonate and sodium carbonate- Calcium carbonate is heated in a kiln to form carbon dioxide and calcium oxide (CaCO3(s)--> CO2(g) + CaO(s) ). The calcium oxide produced is removed to be used for ammonia recovery, and the coke that is present in the kiln produces more carbon dioxide when it is heated. The heat that is emitted causes the calcium carbonate to decompose. Ammonia will then be dissolved in NaCl, a purified brine, and the carbon dioxide is then dissolved in this solution. Chemical equation NaCl(aq) + NH3(g) + H2O(l) + CO2(g) --> H4Cl(aq) + NaHCO3(aq). The reaction is done at a low temperature (0 degrees) so that the sodium hydrogen carbonate precipitates out. Mixture is then filtered, and the sodium hydrogen carbonate is washed then dried and used to make sodium carbonate. After this, the ammonium chloride filtrate is sent to an ammonia recovery plant where the ammonia is recovered and reused. Sodium hydrogen is heated at about 300 degrees and then it decomposes to sodium carbonate and carbon dioxide sodium carbonate is removed then sold, the left over carbon dioxide is reused.
3) Ammonia Recovery- Calcium oxide (from the first step) is dissolved in water to form calcium hydroxide. CaO(s) + H2O(l) --> Ca(OH)2(aq). Ammonium chloride is reacted with this calcium hydroxide which forms calcium chloride and ammonia.The ammonia is reused, but calcium chloride is waste. 2NH4Cl(aq) + Ca(OH)2(s) --> 2NH3(g) + CaCl2(aq) + 2H2O(l)
