Static Equilibrium Theory

Static equilibrium is when all of the particles of the system are at rest and the total force on each particle in permanently zero. It is a perfect balance. Dynamic forces are not acting on the potential energies of the reverse and forward reactions. There is no movement between the reactants and products. It is basically an object that is not moving.
----------------------------------------------------------------------------------------------------------------
Below there is a video that gives you a more physics point of view on static equilibrium.
Terms to know before watching video:
Torque- A measure of how much a force is acting on an object to make the object rotate.
----------------------------------------------------------------------------------------------------------------
Below there is a video that gives you a more physics point of view on static equilibrium.
Terms to know before watching video:
Torque- A measure of how much a force is acting on an object to make the object rotate.
Economic Equilibrium
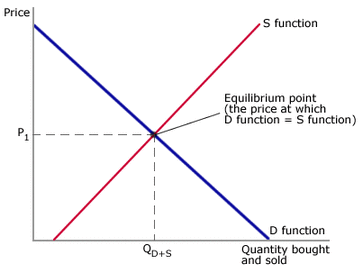
In economics (not in chemistry) is where economics factors such as supply and demand are balanced and in the absence of external influences the (equilibrium) values of economic variables will not change. In other words it is basically a condition or state where in which economic factors are forced to balance. Economic equilibrium may also be defined as the point where supply equals demand for a product – the equilibrium price is where the hypothetical supply and demand curves intersect.
Equilibrium: When supply and demand are equal the economy is said to be at equilibrium. At this point the goods being made is equal to the goods being demanded. This is when everyone is satisfied with the current economic condition. Suppliers are selling all the goods they have produced and consumers are getting all of the goods that they are demanding.
Disequilibrium: Occurs when the supply and demand are not equal. It could be when their is more supply then demand, or more demand then supply. This branches into excess supply and demands.
The picture in the left describes economic equilibrium. The red line (sS function) represents supply. While the blue line represents (D function) the demand. Because they are both equal and balanced they meet in the middle.
Equilibrium: When supply and demand are equal the economy is said to be at equilibrium. At this point the goods being made is equal to the goods being demanded. This is when everyone is satisfied with the current economic condition. Suppliers are selling all the goods they have produced and consumers are getting all of the goods that they are demanding.
Disequilibrium: Occurs when the supply and demand are not equal. It could be when their is more supply then demand, or more demand then supply. This branches into excess supply and demands.
The picture in the left describes economic equilibrium. The red line (sS function) represents supply. While the blue line represents (D function) the demand. Because they are both equal and balanced they meet in the middle.
Equilibrioception

It is also known as a sense of balance. It helps prevent animals and humans from falling over when walking or standing still. Equilibrioception refers to a combination of processes by which an organism can perceive its orientation with respect to gravity.
In humans equibrioception is sensed by the detection of acceleration, this occurs in the vestibular system. (Venstibular system: In the vestibular system equilibrioception is determined by the level of fluid)
Everything has to be balanced to each other and to gravity (equlilbrium!!)
In humans equibrioception is sensed by the detection of acceleration, this occurs in the vestibular system. (Venstibular system: In the vestibular system equilibrioception is determined by the level of fluid)
Everything has to be balanced to each other and to gravity (equlilbrium!!)
Punctuated Equilibrium
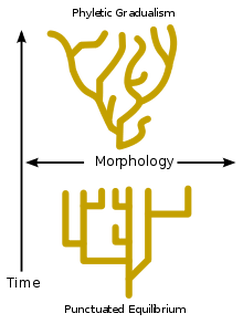
It is a theory in evolutionary biology which proposes that most sexuality reproducing species state will experience little net evolutionary change for the remainder of their history and remain in the same state.
Punctuated equilibrium is a model for discontinuous tempos of change (in) the process of speciation and the deployment of species in geological time.
The image to the left shows you a picture of Phyletic Gradualism (this is when a species slowly adapts to a new species, top picture). The bottom picture shows Punctuated Equilibrium (this is when a species stays continuously the same). The picture shows you the differences between the two. Morphology is a branch of biology dealing with the form and structure of organisms. This includes outward appearance (colour, shape, pattern structure) and the internal parts (organs and bones)
Punctuated equilibrium is a model for discontinuous tempos of change (in) the process of speciation and the deployment of species in geological time.
The image to the left shows you a picture of Phyletic Gradualism (this is when a species slowly adapts to a new species, top picture). The bottom picture shows Punctuated Equilibrium (this is when a species stays continuously the same). The picture shows you the differences between the two. Morphology is a branch of biology dealing with the form and structure of organisms. This includes outward appearance (colour, shape, pattern structure) and the internal parts (organs and bones)
Radioactive/Secular Equilibrium
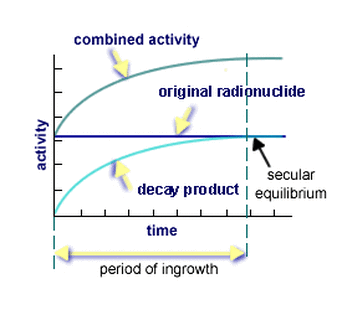
In nuclear physics, secular equilibrium is a situation in which the quantity of a radioactive isotope (an isotope that is radio active) which remains constant because its productive rate is equal to its decay rate. This may get confusing!
Assume that the radioactive material is forming at an almost constant rate. If, at the beginning, no radioactive daughter material is present, no nuclei will be under going transition. As soon as the radioactive material begins to accumulate. As the quantity of radioactive material builds, the activity or transition rate increases until it is equal to the rate of formation as shown below. In other words, radioactive nuclei undergo transitions at exactly the same rate they are forming, and a condition of equilibrium is established.
If your still confused click here
Assume that the radioactive material is forming at an almost constant rate. If, at the beginning, no radioactive daughter material is present, no nuclei will be under going transition. As soon as the radioactive material begins to accumulate. As the quantity of radioactive material builds, the activity or transition rate increases until it is equal to the rate of formation as shown below. In other words, radioactive nuclei undergo transitions at exactly the same rate they are forming, and a condition of equilibrium is established.
If your still confused click here
Vapour-Liquid Equilibrium

It is when a liquid and its vapour (gas form) are at equilibrium with each other, a state when the rate of evaporation is equal to the rate of condensation.
Vapor-Liquid Equilibria is important because it is used in many industrial separation processes. Boiling the mixture produces a partial separation of the components. If the vapor is collected, it can be condensed and boiled again to further change the composition.
At equilibrium, the temperature and pressure of the liquid and vapor phases will be equal, but the compositions will not. There are two main factors that make the vapor and liquid compositions different at equilibrium: the pure component vapor pressures and the nonidealities in the liquid phase.
The picture on the left is a machine known as the Vapour-Liquid Apparatus and is essential for determining the theoretical number of separation stages and other distillation conditions
Vapor-Liquid Equilibria is important because it is used in many industrial separation processes. Boiling the mixture produces a partial separation of the components. If the vapor is collected, it can be condensed and boiled again to further change the composition.
At equilibrium, the temperature and pressure of the liquid and vapor phases will be equal, but the compositions will not. There are two main factors that make the vapor and liquid compositions different at equilibrium: the pure component vapor pressures and the nonidealities in the liquid phase.
The picture on the left is a machine known as the Vapour-Liquid Apparatus and is essential for determining the theoretical number of separation stages and other distillation conditions
Gibbs-Donnan Equilibrium,m
When two solutions of different strengths are separated by a permeable membrane, their concentration on both sides of the membrane equalizes due to diffusion of the solutes as well as the solvent. The effect is named after the American physicist Josiah Willard Gibbs and the British chemist Frederick G. Donnan.
Watch the video down low to see how it works!
Watch the video down low to see how it works!
