History

It was Henry Louis Le Chatelier who is responsible for devising the Le Chatlier Principle. He is French and taught at Sorbonne University
Le Chatelier's Principle

Le Châtelier's Principle states that if you change the conditions of an equilibrium, the equilibrium will shift in a way that minimizes the effects of whatever it is you did.
In other words picture equilibria is like obnoxious little kids. For example, if you yell at a little kid, the kid will change his behaviour to minimize your yelling. Likewise, if you change the conditions of an equilibrium, it will change in a way that partially undoes whatever it is you did to it in the first place.
There are four things that can directly disturb a equilibrium system:
Concentration change, temperature change, pressure and volume change.
In other words picture equilibria is like obnoxious little kids. For example, if you yell at a little kid, the kid will change his behaviour to minimize your yelling. Likewise, if you change the conditions of an equilibrium, it will change in a way that partially undoes whatever it is you did to it in the first place.
There are four things that can directly disturb a equilibrium system:
Concentration change, temperature change, pressure and volume change.
Concentration Change
By adding more products or reactants to the equilibrium system (closed system) it is possible to shift the equilibrium to either favour the products or reactants. When increasing concentration on one side of the equation the equilibrium will shift in the opposite direction.
In the graph to the left, H2 has an increase in concentration, which is why it spikes up before going back to equilibrium. The rate of ammonia will increase which is why it goes up little, but the rate of product will remain the same, returning it to equilibrium. N2 rate of change, goes down (slower) before it return to equilibrium.
In the graph to the left, H2 has an increase in concentration, which is why it spikes up before going back to equilibrium. The rate of ammonia will increase which is why it goes up little, but the rate of product will remain the same, returning it to equilibrium. N2 rate of change, goes down (slower) before it return to equilibrium.
Temperature Change

When energy is either added or removed, the equilibrium shifts to minimize the change in energy. (Depends on whether the reaction is exothermic or endothermic)
The system below is being cooled (losing energy) and is exothermic because the energy is on the product side.
2SO2+O2 ⇌ 2SO3+energy
------------------------------>
The energy is decreasing, so the reactants are making more energy to make up for the loss, making the equilibrium favour the products.
Pressure and Volume Change
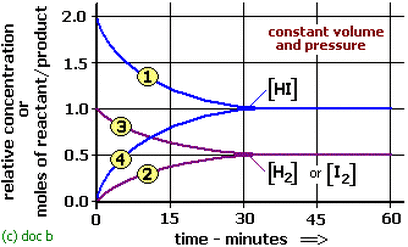
Pressure and volume are inversely proportional (Boyle's Law: P1V1=P2V2). When pressure is increased the volume is decreased. When the volume is increased the pressure is decreases.
When there is a decrease in pressure, there is more volume so the equilibrium will favour the side with more moles:
1N2+3H2⇌ 2NH3+heat
4moles vs 2moles
<--------------------------
When there is a increase in pressure, there is a decrease in volume. The equilibrium will favour the side with less moles:
1N2+3H2⇌ 2NH3+heat
4moles vs 2moles
---------------------------->
When there is a decrease in pressure, there is more volume so the equilibrium will favour the side with more moles:
1N2+3H2⇌ 2NH3+heat
4moles vs 2moles
<--------------------------
When there is a increase in pressure, there is a decrease in volume. The equilibrium will favour the side with less moles:
1N2+3H2⇌ 2NH3+heat
4moles vs 2moles
---------------------------->

